Really? Tell that to Rudolf Clausius who wrote that in as a fundamental part of his equation when formulating the law. 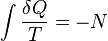 (open system)
(open system)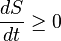 (closed system)
(closed system)
where S is the entropy and t is time.
Yes, and so what?
Simply put, because the law makes a different if the system is an open or closed system because you are introducing outside energy. Furthermore, the concept of entropy in thermodynamics is not identical to the common notion of "disorder". For example, a thermodynamically closed system of certain solutions will eventually transform from a cloudy liquid to a clear solution containing large "orderly" crystals. Most people would characterize the former state as having "more disorder" than the latter state. However, in a purely thermodynamic sense, the entropy has increased in this system, not decreased. The units of measure of entropy in thermodynamics are "units of energy per unit of temperature". Whether a human perceives one state of a system as "more orderly" than another has no bearing on the calculation of this quantity. The common notion that entropy in thermodynamics is equivalent to a popular conception of "disorder" has caused many non-physicists to completely misinterpret what the second law of thermodynamics is really about.