Posted on 02/16/2010 11:40:51 AM PST by Ernest_at_the_Beach
Here’s why it’s possible that doubling CO2 won’t make much difference.
The carbon that’s already up in the atmosphere absorbs most of the light it can. CO2 only “soaks up” its favorite wavelengths of light and it’s close to its saturation point. It manages to grab a bit more light from wavelengths that are close to its favorite bands but it can’t do much more, because there are not many left-over photons at the right wavelengths.

The natural greenhouse effect is real, and it does keep us warm, but it’s already reached its peak performance.
Throw more carbon up there and most of the extra gas is just “unemployed” molecules.
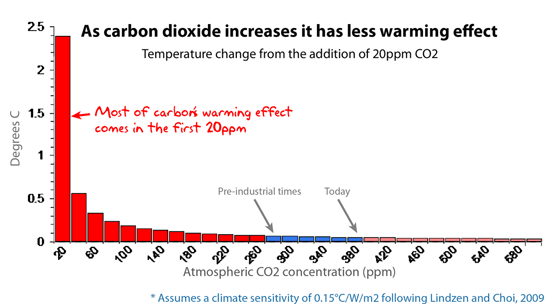
This graph shows the additional warming effect of each extra 20ppm of atmospheric CO2.
AGW says: The climate models are well aware of the logarithmic absorption curve and use it already.
Skeptics say: The models make brutal estimates and many assumptions (guesses). “Lab-warming” doesn’t necessarily translate to “planet-warming”: test tubes don’t have ocean currents, clouds, or rain. The “clouds and humidity” factor is bogglingly complex. For example, high clouds tend to warm the planet but at the same time, low clouds tend to cool it. So which effect rules? Models don’t know but they assume clouds are net-warming. This is not a minor point, the feedback from clouds and humidity accounts for more than half of carbon’s alleged ‘effect’. E’Gad.
AGW says: It’s not 100% saturated.
Skeptics say: True, but meaningless. Log curves never get to “100%”. (So even the air on Venus, which is almost pure CO2, does not absorb 100% of the infra red light). Every CO2 molecule will increase warming by a small amount ad infinitum, but it has less effect than the CO2 that’s already up there.
And the effect is already so small, it’s unmeasurable.
Conclusion: If adding more CO2 to the sky mattered, we would see it in ice cores and thermometers. We don’t. Ergo: Carbon’s effect is probably minor.
When someone pointed out this basic chemistry to me, it resonated, and again I marvelled that something so basic had been carefully not mentioned in this debate. I realize log curves are not something you want to reach out to the public with in detail, but I felt everyone who has done chemistry at university would grasp this point quickly. It explains the paradox: it’s true that carbon has some warming effect, but it’s also true that extra carbon doesn’t have the same effect. Every time alarmists point out that the natural greenhouse effect causes “X degrees of warming” they usually omit to mention that the first 100pm does almost all of that, and no other 100ppm above that will ever do as much. It’s a lie by omission.
The graph in the first printed edition of The Skeptics Handbook is shown below and comes from David Archibald who was the first one to arrange the results in this powerful graphic format. It’s a good graph and he deserves credit for being the one to capture the increasing ineffectiveness of carbon very well. This was calculated (like the top graph) using Modtran, which is a model provided by Spectral Sciences and the US Air Force, and used by researchers around the world.
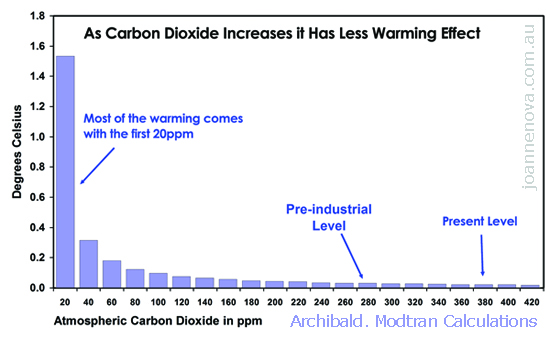
Archibald based his figures on a climate sensitivity calculated by Craig Idso and published in peer reviewed literature (Idso 1998). The graph itself was not published in peer reviewed paper. The top graph above comes from Patrick Michaels and was also constructed on Modtran. It started with a climate sensitivity estimate from Richard Lindzen in his recent ERBE paper (Lindzen and Choi 2009). Useful discussions on the observational backing for a low climate sensitivity is at Friends of Science and also at Niche Modelling.
The two log curves here are not that different, (which is one of the things about a log curve… once you get past the initial slide, it’s all “small” or “smaller”). Overall, both graphs accomplish what I wanted, namely, to show people that the basic effect of carbon dioxide on it’s own dwindles to almost nothing. Sure each extra molecule of carbon makes a little difference, but it becomes less and less so, and there’s a point where it’s irrelevant and unmeasurable. We’re not at that point yet. Even if doubling carbon leads only to a 0.5 degree difference on a global scale, it’s arguably still “measurable” (well… at least theoretically).
My point with this page was not that we could use Modtran to calculate whether there is a crisis due to carbon. I was not so much interested in the exact numbers, as in the shape of the curve. From a science communicators point of view, this is basic science: that additional carbon has less effect. (Can anyone find a school climate education program with this basic chemistry?)
The exponential “hockey-stick” curves of the IPCC et al emphasize just how much difference extra carbon supposedly makes. Few people realize that the exponential rising curves come from feedback factors. (Which are the fatal flaw of the science behind the scare campaign.)
There have been plenty of people who claim the log graph is totally completely utterly all wrong. Desmog tried to claim that Earths atmosphere wasn’t even close to saturated, “look at Venus “. So I did and demolished their point in this reply. (Basically Venus’s atmosphere is 90 times denser than Earths. No wonder it’s hot. It wouldn’t matter what gas was in it’s atmosphere).
Then people come out with irrelevant things, like the Idso paper is “old”, (and so is the theory of gravity), they launch ad hominem attacks on Archibald, and claim the graph is not peer reviewed. The most inflated one is when they claim that the creator of modtran says Archibald misused it. Which really turns out to be merely that the guy who developed the web interface for the modtran model didn’t like the climate sensitivity that Archibald chose (and we are supposed to care?)
The short killer summary: The Skeptics Handbook. The most deadly point: The Missing Hot Spot.
fyi
I want to include the link to a paper Southack gave us...looking for it now.
Falsification Of The Atmospheric CO2 Greenhouse Effects Within The Frame Of Physics
Bookmark.
If anyone makes the argument that CO2 caused the warming on Venus:
The atmosphere of Venus is 90 times thicker than Earth’s and is 96% carbon dioxide, making the atmospheric carbon dioxide concentration on Venus 300,000 times higher than on Earth. Even so, the high temperatures on Venus are only partially caused by carbon dioxide; a major contributor is the thick bank of clouds containing sulfuric acid. Although these clouds give Venus a high reflectivity in the visible region, the Galileo probe showed that the clouds appear black at infrared wavelengths of 2.3 microns due to strong infrared absorption. Thus, Venus’s high temperature might be entirely explainable by direct absorption of incident light, rather than by any greenhouse effect. The infrared absorption lines by carbon dioxide are also broadened by the high pressure on Venus, making any comparison with Earth invalid.
http://brneurosci.org/co2.html
Yup. The issue is the absoption spectra, and CO2 absorbs all the light in its absorption spectra within 10 meters of the ground.
Sry, should be “almost all.” As explained in the article, a tiny tiny tiny little bit isn’t, and increasing CO2 in the air would capture a tiny tiny part of that. We’re talking way below 1%, though, which for all practical terms is none...
If I’m not mistaken Mars has an atmosphere of 96% carbon dioxide and they’re freezing their a$$ off.
Great article and CO2 is also not a stable molecule but a reactive one and undergoes many reactions and changes into other chemical species.
The natural greenhouse effect is real, and it does keep us warm,
As I understand the paper...see link at #4....
The statement is essentially not true...
The natural greenhouse effect of CO2 is also negligible. The most powerful greenhouse element in our atmosphere is naturally occuring water vapor which accounts for over 95% of what is floating around in our atmosphere. Without it we would be living in an ice age.
Without a natural greenhouse effect, the average temperature on Earth would be about zero degrees (F), not the present 57 degrees.
So the natural greenhouse effect is real. But its mostly due to water vapor; CO2 accounts for only a few of those 57 degrees.
This article is provided as a public service by the Geophysical Institute, University of Alaska Fairbanks, in cooperation with the UAF research community. Dr. Sue Ann Bowling is an Associate Professor of Physics at the Institute.
**************************
If you've ever heard an explanation of how a greenhouse works, it was most likely based on the differing transparency of glass to solar and thermal infrared radiation. Glass is transparent to most of the wavelengths of solar radiation, but is effectively opaque to the much longer (thermal infrared) wavelengths emitted by the plants and soil inside the greenhouse. Solar radiation can get into the greenhouse, where it is absorbed by and heats whatever is inside the greenhouse. The longer wavelengths emitted by the heated surfaces cannot get out through the glass, however, so heat keeps building up -- at least that's how the conventional explanation goes. Polyethylene greenhouses, however, seem to work just about as well as glass ones -- and polyethylene is nearly as translucent to thermal infrared radiation as it is to solar radiation. So why does a greenhouse get so hot on a sunny day?
Whether bare or covered by a greenhouse, the ground absorbs radiation from the sun and heats up. The increase in temperature is conducted to the air next to the earth; that air then warms and expands, thus becoming less dense than the air higher up. The lighter air rises, allowing cooler and denser air to take its place at the surface and absorb more heat from the warmed ground. Thus the radiation absorbed by the ground goes into heating a deepening layer of air. Above open ground on a sunny day in summer, the heated layer of air may easily be a mile or more deep, and since the warming is spread over such a large mass (a deep layer of air plus a very thin layer of soil and vegetation), the temperature rise is diluted by the sheer amount of stuff that must be heated. (Think of how slowly the temperature rises in a large kettle full of water which is set on a hot stove for five minutes.)
In a greenhouse, this mixing is confined to the layer of air trapped under the roof, so there is a much smaller mass to be heated. Essentially, the large kettle full of water has been replaced by one with half an inch of water on its bottom, and as a result the water will warm up much faster. This also explains why ventilation is so important in keeping a greenhouse from overheating. (A closed car in the sun heats up due to the same mechanism.)
Ironically, the notorious "greenhouse effect" really does work the way a gardener's greenhouse was (inaccurately) thought to operate: carbon dioxide, methane, water vapor and other such gasses in the atmosphere let the solar radiation pass to the earth's surface but impede the reradiation of thermal infrared wavelengths back to space.
The old explanation of how a greenhouse works also has some relevance in explaining why the nighttime temperatures in a greenhouse are warmer than those in the unprotected garden. The stored heat of the day continues to be radiated by the ground after the sun goes down, so the ground and plants cool. Cooling of the air near the ground does not spread through the atmosphere, as the cooled air is denser than that above. But in the greenhouse, the cooling starts from the greenhouse roof. The stored heat from the full height of the greenhouse is available overnight, and the plants never get much colder than the air. The process can be made more efficient, and at the same time the overheating during the day minimized, by increasing the thermal mass of the greenhouse, perhaps by storing black-painted barrels of water under the plant benches.
The night-time radiative effect works even with a polyethylene greenhouse, because the first thing that happens when the air starts to cool is that some of the water from the moist air of the greenhouse condenses on the inside of the greenhouse roof. Water, like glass, is opaque to thermal infrared, so the polyethylene and glass greenhouses are effectively identical in their night-time behavior -- provided you've remembered to water the greenhouse plants!
That article makes no sense to me. I think it is debunking an exact parallel between the way a glass greenhouse works and the way the atmosphere works. Also, the issue is water v. C02. Why does it get so cold in the desert at night but not on a cloudy day in manhattan? Radiant longwave cooling from the earth’s surface out to space being blocked by atmospheric gases is obviously a huge part of the equation.
I think it’s real but miniscule. In daylight, CO2 absorbs and at night it re-emits. I’m thinking of humans in infrared vision scopes at night. What’s emitted from warm blooded people at night is huge compared to the atmosphere.
In organic chemistry synthesis, ultraviolet light often supplies the energy of activation, i.e. the energy needed to get a reaction started. They’re looking for catalysts that can use visible light for the energy of activation. Maybe in vegetation the chloroplasts can use weaker infrared radiation for the energy of activation.
Just my 2 cents.
The natural greenhouse effect of CO2 is also negligible.
Examine this table to give you perspective on CO2 concentration:
http://www.freerepublic.com/focus/news/2451661/posts?page=34#17
Examine this table
Thanks again
Disclaimer: Opinions posted on Free Republic are those of the individual posters and do not necessarily represent the opinion of Free Republic or its management. All materials posted herein are protected by copyright law and the exemption for fair use of copyrighted works.