
The trial was completed by Professor Didier Raoult, based at infectious diseases institute, IHU Méditerranée Infection in Marseille
Posted on 03/18/2020 8:06:51 PM PDT by SeekAndFind

The trial was completed by Professor Didier Raoult, based at infectious diseases institute, IHU Méditerranée Infection in Marseille
A renowned research professor in France has reported successful results from a new treatment for Covid-19, with early tests suggesting it can stop the virus from being contagious in just six days.
Professor Didier Raoult from infection hospital l'Institut Hospitalo-Universitaire (IHU) Méditerranée Infection in Marseille (Bouches-du-Rhône, Provence-Alpes-Côte d'Azur), published a video explaining the trials on Monday March 16.
Professor Raoult is an infectious diseases specialist and head of the IHU Méditerranée Infection, who has been tasked by - and consulted by - the French government to research possible treatments of Covid-19.
He said that the first Covid-19 patients he had treated with the drug chloroquine had seen a rapid and effective speeding up of their healing process, and a sharp decrease in the amount of time they remained contagious.
Chloroquine - which is normally used mainly to prevent and treat malaria - was administered via the named drug, Plaquenil.
The treatment was offered to 24 patients, who were among the first to become infected in the south east of France, and who had voluntarily admitted themselves to hospital for the process.
Patients were given 600mcg per day for 10 days. They were closely monitored, as the drug can interact with other medication, and cause severe side effects in some cases.
Professor Raoult said: “We included everyone who was in agreement [to be treated], which was almost everyone. Two towns in the protocol, Nice and Avignon, gave us [infected] patients who had not yet received treatment.
“We were able to ascertain that patients who had not received Plaquenil (the drug containing hydroxychloroquine) were still contagious after six days, but of those that had received Plaquenil, after six days, only 25% were still contagious.”
Chloroquine phosphate and hydroxychloroquine have previously been used to treat coronavirus patients in China, in ongoing Covid-19 clinical trials.
Kaletra, a US-based antiviral drug normally used to treat HIV, is another medicine that is being tested in the fight against Covid-19.
A new academic study, published on Friday March 13 by US scientific researchers, also said that chloroquine appeared to be an effective treatment, and appears to align with the findings in France.
It said: “Use of chloroquine (tablets) is showing favorable outcomes in humans infected with Coronavirus including faster time to recovery and shorter hospital stay…
“Research shows that chloroquine also has strong potential as a prophylactic (preventative) measure against coronavirus in the lab, while we wait for a vaccine to be developed.
“Chloroquine is an inexpensive, globally available drug that has been in widespread human use since 1945 against malaria, autoimmune and various other conditions…[it] can be prescribed to adults and children of all ages.
"It can also be safely taken by pregnant women and nursing mothers [and] has been widely used to treat human diseases, such as malaria, amoebiosis, HIV, and autoimmune diseases, without significant detrimental side effects.”
Researchers worldwide are continuing to work on developing a vaccine against Covid-19.
So far, no country - nor the World Health Organisation (WHO) - has officially published treatment measures against Covid-19, but in China and South Korea, guidelines already outline the use of chloroquine as an “effective treatment”, the study report said.
Kevin McCullough
@KMCRadio
·
9h
Physicians can prescribe immediately!
READ THIS STUDY:
https://docs.google.com/document/d/e/2PACX-1vTi-g18ftNZUMRAj2SwRPodtscFio7bJ7GdNgbJAGbdfF67WuRJB3ZsidgpidB2eocFHAVjIL-7deJ7/pub
While unpublished yet, the first patient trial of Plaquenil for COVID-19, conducted in Marseilles, France, has reported encouraging early results. The trial, announced in a video on Monday, was led by Didier Raoult, a physician-scientist who has published on microbes for several decades.
According to a draft article by Raoult’s team and obtained by me, 36 patients were enrolled in the trial, including 16 infected controls and 20 treated patients. The test group was given 600 mg daily of Plaquenil, which is on the WHO’s List of Essential Medicines and has been used for malaria, rheumatoid arthritis and lupus.
At day three, the study reported, 50 percent of the treated group turned from positive to negative for the COVID-19 virus. By day six, 70 percent tested negative.
As intriguing, of the 20 test patients, six who were treated with both Plaquenil and the antibiotic azithromycin did even better, the team reported. Five of the six, or 83 percent, tested negative at day three. All six, 100 percent, tested negative at day six.
Meanwhile, the control patients largely stayed sick longer. On days three and six, just 6.3 percent and 12.5 percent, respectively, tested negative. The research paper did not include how they were alternatively treated.
Raoult’s findings prompted the French Minister of Health Tuesday to approve expanded treatment trials, with one commencing in Lille, according to France 3, a public television station.
Sounds good.
Spread rates have been pretty slow in Africa and Latin America. Could it be that enough patients take chloroquinine for malaria or malaria prophylaxis that there is a large body of people who can neither get or transmit because they are on this drug?
If anyone can teach the virus to “surrender”... go France! :)
We're an independent group of scientists and physicians working on an open-data clinical trial for prevention of COVID-19, through the use of hydroxychloroquine in combination with other therapeutic agents.
Unlike a typical commercial drug trial, our objective is to share trial data with the public* and health-care professionals as close to real-time as possible (with a reasonable level of data quality assurance).
Given the rapidly spreading coronavirus pandemic, we're looking for every possible means to fast-track the effort.
* Data will be de-identified to preserve participants' privacy and conform with regulatory requirements.
Objective: Evaluate the efficacy of hydroxychloroquine in the prevention of COVID-19 infection.
Current Phase: We're first focusing on a cohort study of healthy medical professionals.
Status: Active / Recruiting
Join the study: If you're a front-line healthcare worker (physician, nurse, etc.), and willing to participate in the trial (or already taking hydroxychloroquine), please send us an email.
Future phase: Case-control study of hydroxychloroquine in the prevention of COVID-19. Stay tuned.
If you're interested to support or partner on regulatory front, clinical trial, or funding, please send us an email.
A recent well controlled clinical study conducted by Didier Raoult M.D/Ph.D, et. al in France has shown that 100% of patients that received a combination of HCQ and Azithromycin tested negative and were virologically cured within 6 days of treatment.
In addition, recent guidelines from South Korea and China report that hydroxychloroquine and chloroquine are effective antiviral therapeutic treatments for novel coronavirus.
A therapeutic agent that prevents infection with novel coronavirus is highly desirable--especially for persons with high-risk exposure (e.g healthcare professionals) as well as persons with comorbidities (heart disease, diabetes, etc) and compromised immune systems. Ground-breaking in vitro studies demonstrate potential efficacy of hydroxychloroquine as a prophylactic for novel coronavirus infection in primate cells.
Note: Hydroxychloroquine (brand name Plaquenil) is an inexpensive, globally available drug (tablet) that was approved for widespread medical use since 1955. It is commonly used today to treat malaria, systemic lupus erythematosus and rheumatoid arthritis.
Gregory J. Rigano, Esq
Mr. Rigano is an Advisor to the Stanford University School of Medicine SPARK Translational Research Program. He's led a biotech firm for the past five years in research and clinical evaluation of Chloroquine in various diseases.
Gregory has provided counsel to over $1 billion in transaction volume at global scale with a strong focus on the sciences involving multi-national corporations and the federal government. Gregory’s experience includes advancing various pharmaceutical assets through laboratory, animal, formulation, manufacturing, clinical trials (Phase I - III) as well as commercialization. Mr. Rigano received his Juris Doctor degree from Hofstra University, and studied at Johns Hopkins University.
Didier Raoult, MD & Ph.D
Didier Raoult created the Rickettsia Unit at Aix-Marseille University. Since 2008, Dr. Raoult has served as the director of URMITE (Research Unit in Infectious and Tropical Emergent Diseases), collaborating with CNRS (National Center for the Scientific Research), IRD (Research for the Development Institute), INSERM (National Institute of Health and Medical Research) and Aix Marseille University. His laboratory employs more than 200 people, including nearly 100 active researchers who publish between 250 and 350 papers per year and have produced over 50 patents.
RE: Spread rates have been pretty slow in Africa and Latin America. Could it be that enough patients take chloroquinine for malaria or malaria prophylaxis that there is a large body of people who can neither get or transmit because they are on this drug?
The warm and hot weather helps too.
“Spread rates have been pretty slow in Africa and Latin America. Could it be that enough patients take chloroquinine for malaria or malaria prophylaxis that there is a large body of people who can neither get or transmit because they are on this drug?”
Sounds plausible that at minimum it could be a factor.
Dude, you win the internets for the day. That made me laugh hard.
Elon Musk recently suggested the same drug.
SEE POST #6 ABOVE.
When the predictive number is under 0.5% for how well a study can be replicated by others, that is considered GOLDEN. If predictive number is .01%, you are 50 times better of than what is considered golden.
The said study by Dr. Raoult is .0025% in its p value ( predictive value ). This means that this drug is nearly 100% effective in killing the coronavirus (better than 99% ) and this is also the chances that any scientist who follow the same protocol will replicate the results again and again.
What does this mean?
Hydroxychloroquine are already in every pharmacy shelf in this country, so the drug will NOT BE Expensive.
They also discovered that when you mix Hydroxychloroquine with azithromycin, they have the best results on Coronavirus patients. If this is true, and studies show that it is, the goal should be to get these two drugs to as many doctors treating Covid-19 patients as possible.
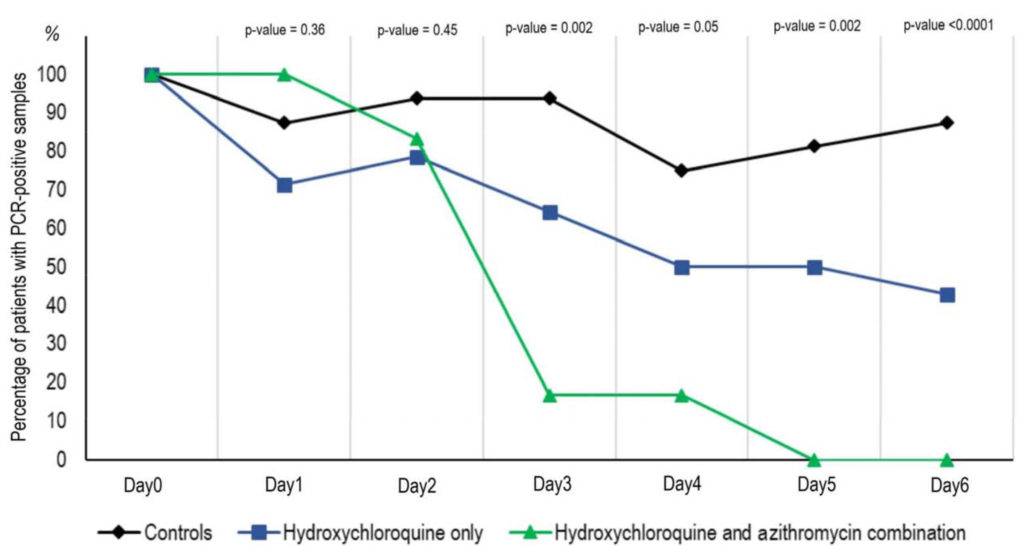
The above graph shows the Percentage of patients with PCR-positive nasopharyngeal samples from inclusion to day 6 post-inclusion in COVID-19 patients treated
with hydroxychloroquine only, in COVID-19 patients treated with hydroxychloroquine and azithomycin combination, and in COVID-19 control patients.

This was on Tucker tonight... if confirmed markets are going to skyrocket.
I remember Chloroquine well.
By the way, Hydrochloroquine is in the VA formulary.

I saw an explanation of this on MedCram a few days ago. In that, this drug was paired with zinc. The drug helped the cells grant entry to the zinc so that cells could use it.
Nice graph. At worst people had better give a damned good explanation for dummies like me if they don’t aggressively pursue this and soon.
Disclaimer: Opinions posted on Free Republic are those of the individual posters and do not necessarily represent the opinion of Free Republic or its management. All materials posted herein are protected by copyright law and the exemption for fair use of copyrighted works.